 Evolution
Evolution
 Intelligent Design
Intelligent Design
More Marvels in the Molecular Machine Menagerie
As a plausible explanation of life’s complexity, Darwinian thinking emerged when cellular biology was a great blur. As what we know about cells and their contents has dramatically sharpened in detail and focus, orthodox evolutionary thinking correspondingly fades in its persuasiveness. That’s a lesson of what we wrote yesterday on molecular machines (“Molecular Machine Menagerie Brightens“), but the latest news on that theme can’t be encompassed by a single article, or two. Therefore we move on to automated security agents, linemen, recyclers, thermostats, assemblers, inspectors, bodyguards, and more.
Addendum on ion channels. Here’s another new paper about voltage-gated sodium channels, called Navs. In humans, these are involved in sensory neurons as well as heart and brain cells, but even microbes have them. We’ll share one quick quote from the paper in Nature Communications:
The cycling of Navs through open, closed and inactivated states, and their closely choreographed relationships with the activities of other ion channels lead to exquisite control of intracellular ion concentrations in both prokaryotes and eukaryotes. [Emphasis added.]
Skilled linemen. Researchers at the Technical University of Darmstadt were surprised to learn that the machines that repair double-stranded breaks in DNA are “far more complex than previously assumed.” For instance, “The ends of breaks in the double helix are not just joined, they are first changed in a meticulously choreographed process so that the original genetic information can be restored.” Interesting that both the above entries refer to choreography — a great design word suggesting irreducible complexity — augmenting the impact with the adjectives exquisite and meticulous.
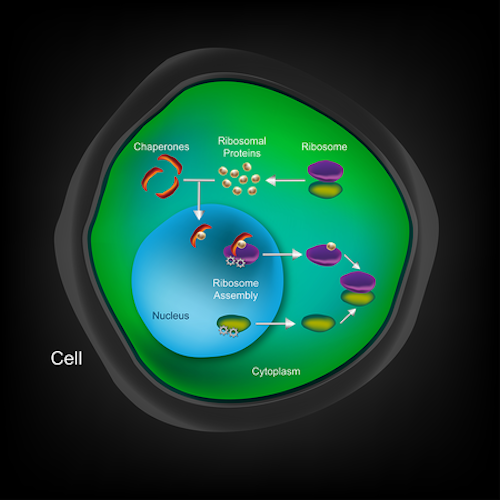
No-nonsense chaperone. You can see another machine’s structure in 3-D at Caltech News, where the article states, “Protein chaperone takes its job seriously.” What is it? It’s a ribosomal protein’s secret service bodyguard, essentially:
For proteins, this would be the equivalent of the red-carpet treatment: each protein belonging to the complex machinery of ribosomes — components of the cell that produce proteins — has its own chaperone to guide it to the right place at the right time and protect it from harm.
The particular protein they studied, named L4, has a chaperone that fits tightly like a hand and glove. When the protein is produced in the nucleus, the chaperone takes it on a long trip out the nuclear pore and into the cytoplasm, where it has to be fitted into the ribosome at the right place and time. Along the way, the chaperone protects its client from being chopped up by the “protein-shredding machinery.” This article is loaded with amazing facts. For example,
Building ribosomes is a formidable undertaking for the cell, involving about 80 proteins that make up the ribosome itself, strings of ribosomal RNA, and more than 200 additional proteins that guide and regulate the process. “Ribosome assembly is a dynamic process, where everything happens in a certain order. We are only now beginning to elucidate the many steps involved,” says [André] Hoelz.
That’s a picture of choreography again. One more little factoid if you’re not impressed yet: “More than a million ribosomes are produced per day in an animal cell.” This is one big ballet!
The Shredder. The article above mentioned “protein-shredding machinery,” so let’s see what’s new about that. A European team publishing in the Proceedings of the National Academy of Sciences has learned that the 26S Proteosome, “a large multisubunit complex that executes the degradation of intracellular proteins marked for destruction,” contains an “engine” with moving parts. This engine “unfolds and translocates substrates into the 20S core particle” where the protein is shredded, allowing its amino acids to be recycled. How does the machine know what to recycle? Mistakes could be disastrous, harming working proteins. For proper identification, other machines “tag” the trash with ubiquitin molecules. The proteasome checks the tags before letting the protein into the recycling bin. We’ve all seen the trucks that lift trash cans into big bins and shake them. Something like that goes on in the cell:
Here, we report cryo-EM structures of the yeast 26S proteasome in the presence of different nucleotides and nucleotide analogs, revealing the existence of four distinct conformational states. These structures elucidate the conformational changes underlying substrate translocation and their coupling with gate opening.
This is no clunky trash truck. Processing involves multiple steps, gate openings, twists and turns in a specific sequence, all requiring ATP for energy. “We assume that the cycle continues until the substrate translocation process is finished,” they say in conclusion. “Our structures favor a model in which the hydrolysis cycle occurs in a sequential order around the ring rather than in a stochastic manner.” Interestingly, one of the positions they call the “lockwasher conformation.” There could well be additional dance steps of this cellular robot that haven’t been discovered yet.
Inspector-Ejectors to the rescue. In the ribosome, messenger RNAs are translated into proteins. What if the mRNA has a typo? What if it lacks a stop codon? The resulting protein could be damaged, or even dangerous. “The ability to dispose of proteins that are either aberrant or (in the worst case) toxic is fundamental to a cell’s survival, says news from Charité University of Medicine in Berlin. Researchers describe “rescue proteins” that patrol ribosomes, providing the necessary quality control on the assembly line. The next question is: how do they recognize errors?
Using cryo-electron microscopy to study the structure of such ribosome-mRNA complexes, the researchers were able to show the manner in which special rescue proteins (Dom34 and Hbs1) recognize such stalled ribosomes, thereby initiating the splitting of the arrested complex and the degradation of the faulty mRNA. The rescue proteins recognize arrested ribosomes by detecting, and binding to, conserved locations normally occupied by mRNA. This direct competition-based approach ensures that only ribosomes with aberrant mRNAs are targeted.
The Stapler. Briefly, an article from Ludwig Maximilian University of Munich describes protein machines that attach to mRNAs as they exit the nucleus and stabilizes them for transport. “We were surprised to see that the RNA is not only recognized by these proteins, they also force it to adopt a new form. They staple it together, so to speak.” Then the motor proteins “take the mRNA train,” carrying the passenger down the cell’s “railway lines,” the article says picturesquely.
Pressure thermostat. How do epithelial tissues maintain the right number of cells? Researchers at the University of Utah wanted to know. First, they found that simple mechanical tension affects the balance of birth and death. When cells get too crowded, internal pushing forces lead some cells to pop out of the tissue and die, undergoing apoptosis. When cells get too sparse, they pull on each other, triggering cell division and the creation of new cells to fill in the gaps. But then, they discovered a protein machine responsible for this balance. It’s called Piezo1, named undoubtedly for its mechanosensitive nature, like certain crystals that can spark when compressed. Piezo1 acts like a “thermostat” on both sides of the cell, they found.
Just like a thermostat regulates both heat and cold, it makes sense to have one sensor measuring crowding and stretch. If there were two separate regulators, things could get out of hand fairly quickly if one sensor breaks.
Fancy footwork. Did you know some cells have feet? Cells in your immune system, for instance, create about a hundred “podosomes” (foot bodies) to move quickly to their sites of operation. The podosomes secrete proteins that degrade the extracellular matrix, allowing the immune cells to slip through crowded tissues. Researchers at the National University of Singapore wanted to learn more about how cells form these little feet. What they found was too complex to describe in detail here, but it involves multiple proteins that form rings, switches and controllers, with the aid of those myosin motors we learned about last time.
The propeller. We lack space to describe the helical zipper (Science Daily), the DNA surgeon (Phys.org) and other fascinating machines, but our mini-tour of the molecular machine menagerie wouldn’t seem complete without some news on the iconic bacterial flagellum that was so influential in the intelligent design movement. Two recent papers shed more light on the flagellum, both from Osaka University, an institution that has taken the lead on elucidating this propeller’s physical secrets in great detail. One news item from Osaka University explores how pH in the system affects energy when the cell extrudes proteins out of the protein to build the machine. Tiny pH microprobes allowed the team to “propose that the export apparatus uses both ATP hydrolysis as well as H+ differentials to achieve protein export.”
The other paper, published by Nature Communications, explores the rod and hook regions of the flagellum. Osaka researchers found that “identical folds” in subunit proteins FlgE and FlgG are “used for distinct mechanical functions” of the rod and hook, which are directly connected to each other. Though these two proteins share 39 percent sequence identity, they have distinct properties: the rod is straight, but the hook is flexible, allowing it to bend as a universal joint. “While these two structures have the same helical symmetry and repeat distance and nearly identical folds of corresponding domains, the domain orientations differ by ∼7°, resulting in tight and loose axial subunit packing in the rod and hook, respectively, conferring the rigidity on the rod and flexibility on the hook,” they explain. “This provides a good example of versatile use of a protein structure in biological organisms.” That sounds like a good design.
As we said in yesterday’s post, there’s nothing like investigating machines in detail to reinforce the conviction that cells are intelligently designed; they could not have emerged by blind processes of random mutations and natural selection. Some of these machines, when mutated, result in devastating diseases, like ALS and cancer. Little do we know how much our lives depend on precise, reliable action of actual machines with moving parts on the nanoscale that bear uncanny resemblances to machines we know on the human scale: trash compactors, inspectors, propellers, and much more.
In Unlocking the Mystery of Life, Jed Macosko said there were “a host of machines” in the cell, as many as there are functions in the human body. Here in 2017, 15 years later, we can see that was no exaggeration. The fuzzier glimpses of machines that turned Michael Behe into an advocate of intelligent design still have the power to inspire a new generation of young scientists — all the more so with the increasing resolution of advanced imaging techniques.
Image: Cell with chaperone proteins, by Huber and Hoelz via Caltech.
