Intelligent Design
Intelligent Design
How Microbes Make Earth Habitable
Several recent discoveries point to amazing connections between global forces and Earth’s tiniest creatures that maintain the habitability of our planet. Consider some examples and think about the implications.
Atmospheric Requirements
First, let’s talk about the atmosphere. As David Klinghoffer reported here last month at Evolution News, it appears we can dispense with the notion that a rise in oxygen caused the Cambrian explosion. That paper in PNAS suggests that “O2 levels capable of supporting animal physiology were present more than 500 million years before the appearance of animals.” We know that oxygen is necessary for complex life. This paper says it was already here long before complex multicellular animals appeared.
Less recognized is nitrogen. Elizabeth Howell wrote in NASA’s Astrobiology Magazine back in 2014 that oxygen could fool the search for life, but this year she wrote in the same Astrobiology Magazine that nitrogen “may be a sign of habitability.”
We might commonly think of Earth as having an oxygen-dominated atmosphere, but in reality the molecule makes up only a fifth of our air. Most of what surrounds us is nitrogen, at 78 percent. Astrobiologists are beginning to see nitrogen — and not just oxygen — as a key indicator of a planet’s habitability. Nitrogen is essential for life on Earth and could signal an atmosphere thick enough to stabilize liquid water on a planet’s surface, fundamental to creating habitable conditions. [Emphasis added.]
Nitrogen and oxygen may not be sufficient for life (Saturn’s moon Titan, for instance, has plentiful nitrogen), but they are both necessary. A leading source of free oxygen is photosynthesis in autotrophic microbes; and only microbes have the know-how to “fix” nitrogen by dismantling the triple bonds of atmospheric nitrogen into ammonia and other useful compounds. As we will see, microbes play a major role in maintaining the balance other essential atmospheric elements, too. Without that balance, complex life could not exist.
Nitrogen-Fixing Bacterium Does Solo Performance
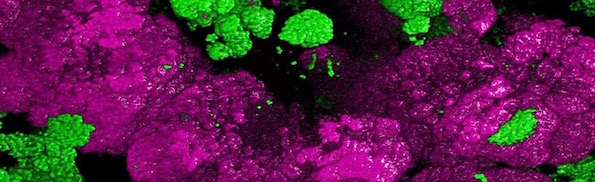
Since we reported about the anammox process last fall, a major discovery has been made about nitrogen-fixing bacteria. Nature reported the discovery of a bacterium that can do complete nitrification.
Nitrification, the oxidation of ammonia via nitrite to nitrate, has always been considered to be a two-step process catalysed by chemolithoautotrophic microorganisms oxidizing either ammonia or nitrite. No known nitrifier carries out both steps, although complete nitrification should be energetically advantageous. This functional separation has puzzled microbiologists for a century. Here we report on the discovery and cultivation of a completely nitrifying bacterium from the genus Nitrospira, a globally distributed group of nitrite oxidizers. The genome of this chemolithoautotrophic organism encodes the pathways both for ammonia and nitrite oxidation, which are concomitantly activated during growth by ammonia oxidation to nitrate. Genes affiliated with the phylogenetically distinct ammonia monooxygenase and hydroxylamine dehydrogenase genes of Nitrospira are present in many environments and were retrieved on Nitrospira-contigs in new metagenomes from engineered systems. These findings fundamentally change our picture of nitrification and point to completely nitrifying Nitrospira as key components of nitrogen-cycling microbial communities.
We’ll have to add a new term to our vocabulary now: comammox or “complete ammonia oxidizer”; see the summary from the University of Vienna. An article from from the same source describes the “incredibly exciting moment” when they found the “long-sought comammox organism” 1,200 meters deep in a Russian oil exploration well. A bigger surprise came when they realized “comammox was everywhere but was overlooked.”
Electric Sediment Buffers Ocean
Life began in the oceans, but without bacteria, it might not survive. According to surprising findings announced in Current Biology:
Animals at the bottom of the sea survive oxygen depletion surprisingly often, and a new study identifies cable bacteria in the sediment as the saviors. The bacterial electrical activity creates an iron ‘carpet’, trapping toxic hydrogen sulfide.
The paper’s title is, “Electrical cable bacteria save marine life.” These “key organisms” link up into electrical “wires” in the top layers of ocean sediment, carrying electrons from sulfide oxidation up into the ocean water, where further reactions form an “iron firewall” that protects the ocean from a buildup of toxic hydrogen sulfide. This buffering of the ocean against harmful conditions even undergoes a seasonal cycle:
Essentially, the cable bacteria manage a sulfide storage facility made of reactive iron, where sulfide is stowed away during oxygen-poor periods and regained for efficient energy metabolism when oxygen returns. Seitaj et al. propose that the control of sulfide mediated by cable bacteria, which they observed at Grevelingen, could be widespread and may explain many cases where oxygen depletions are not followed by release of hydrogen sulfide and mass mortality of marine life.
Plankton Maintain Carbon Cycle
Carbon dioxide levels in the atmosphere are an important concern to climate scientists. It’s been known for a long time that plankton in the surface ocean waters play a big role in the carbon and oxygen cycles. Researchers at MIT took a closer look at certain members of the plankton community called “mixotrophs” — those that can feed by photosynthesis and by consuming prey. They realized that “these microscopic, mixotrophic organisms may have a large impact on the ocean’s food web and the global carbon cycle.”
The MIT scientists note that mixotrophs have been overlooked in climate models. As these heavier plankton die, they take their carbon down into the seafloor sediments with them. Consequently, “mixotrophic organisms may make the ocean more efficient in storing carbon, which in turn enhances the efficiency with which the oceans sequester carbon dioxide.” Without mixotrophs, there would likely be more carbon dioxide in the atmosphere — and we all know what that means. One wonders if the earth would become less habitable over time if it were not for the planet-wide function performed by these tiny organisms.
Diatoms Promote Diatomic Oxygen
One fifth of the oxygen in our atmosphere comes from one-celled algae called diatoms. These incredible organisms, known for the variety of their beautiful silica houses (called tests) inhabit the oceans and fresh waters of the planet in vast numbers. Although Michael Denton doesn’t discuss them in his new book Evolution: Still a Theory in Crisis, he well could have pointed to their beautiful shells as non-adaptive forms inexplicable by natural selection. Why would an organism need to create triangles, rods, and five-pointed stars with intricate etched patterns on transparent glass? Every time a diatom divides, the daughter cells recreate the elegant shells of their particular species.
To do their important work for the global atmosphere, they need to find silicon. An exciting new discovery was announced by Friedrich Schiller University Jena. Diatoms can apparently “smell” silicate minerals, and migrate to where they are plentiful. In a petri dish, scientists watched biofilms of diatoms move two micrometers a second toward silicate sources, but away from toxic minerals. How they do this is unknown, but it was clear to them that “The algae have to search their environment for the building material” (italics in original). The results of their study are published in Nature Communications.
If diatoms lacked the ability to seek out and find what they need, the planet might have reduced oxygen, decreasing its ability to support complex life. Thus our pun on “diatoms” (Greek “cut in two”) and “diatomic oxygen” (“two atoms,” O2) — two unrelated words about pairs of things that interact in a surprising way.
Microbes make earth a nice place to live. There’s more to say on that theme, and we’ll do so in an additional post coming up.
Image: Nitrospira bacteria, by Holger Daims via University of Vienna.