 Evolution
Evolution
 Intelligent Design
Intelligent Design
An "Exquisitely Designed" Enzyme that Maintains DNA Building Blocks
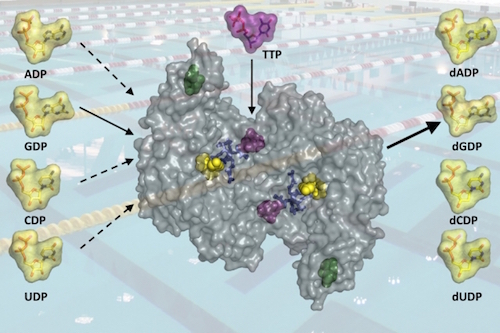
It’s molecular machine time, and today we’ll be looking at a particularly amazing one. It’s essential, it’s “evolutionarily ancient,” and it’s unique. This machine, named ribonucleotide reductase, or RNR for short, is a beauty. News from MIT explains why your life depends on this machine:
Cell survival depends on having a plentiful and balanced pool of the four chemical building blocks that make up DNA — the deoxyribonucleosides deoxyadenosine, deoxyguanosine, deoxycytidine, and thymidine, often abbreviated as A, G, C, and T. However, if too many of these components pile up, or if their usual ratio is disrupted, that can be deadly for the cell.
A new study from MIT chemists sheds light on a longstanding puzzle: how a single enzyme known as ribonucleotide reductase (RNR) generates all four of these building blocks and maintains the correct balance among them. [Emphasis added.]
The image shows the complex machine with active sites that precisely fit the four different building blocks. Its job is to take the ribonucleotides that make up RNA and turn them into the deoxyribonucleotides that make up DNA.
“There’s no other enzyme that really can do that chemistry,” she says. “It’s the only one, and it’s very different than most enzymes and has a lot of really unusual features.“
In order for the machine’s moving parts to work, one of several “effector molecules” has to fit into its special spot, like a key that opens a latch. This causes the enzyme to open up a “distant” active site and let the appropriate RNA building block in. Then, the enzyme latches it into place for its operation.
Depending on which of these effectors is bound to the distant regulatory site, the active site can accommodate one of the four ribounucleotide substrates. Effector binding promotes closing of part of the protein over the active site like a latch to lock in the substrate. If the wrong base is in the active site, the latch can’t close and the substrate will diffuse out.
“It’s exquisitely designed so that if you have the wrong substrate in there, you can’t close up the active site,” Drennan says. “It’s a really elegant set of movements that allows for this kind of molecular screening process.”
This four-in-one machine takes on four different shapes depending on whether the RNA nucleotide is A, G, C, or U. It sends out DNA’s four, A, G, C, and T with the appropriate sugar deoxyribose instead of ribose (replacing an OH radical with a hydrogen atom, a “reduction” reaction). But that’s not all this amazing machine does:
The effectors can also shut off production completely, by binding to a completely different site on the enzyme, if the pool of building blocks is getting too big.
RNR is a multi-tool if there ever was one. The effectors, the substrates and the active sites are all closely matched to the operation at hand, whether generating more DNA building blocks or regulating their supply in the cell. A paper from the Annual Review of Biochemistry (2007) puts it this way:
An intricate interplay between gene activation, enzyme inhibition, and protein degradation regulates, together with the allosteric effects, enzyme activity and provides the appropriate amount of deoxynucleotides for DNA replication and repair.
A lot of new building blocks are needed for repair and replication. You can see why this enzyme is essential for a cell when it divides or suffers stress.
The news item mentioned a latch, but another paper from 2015 in the Journal of Biological Chemistry speaks of a switch mechanism:
Ribonucleotide reductase (RNR) catalyzes the reduction of ribonucleotides to the corresponding deoxyribonucleotides, which are used as building blocks for DNA replication and repair. This process is tightly regulated via two allosteric sites, the specificity site (s-site) and the overall activity site (a-site). The a-site resides in an N-terminal ATP cone domain that binds dATP or ATP and functions as an on/off switch, whereas the composite s-site binds ATP, dATP, dTTP, or dGTP and determines which substrate to reduce.
It’s like a surgical robot that has a clamp with an on-off switch. The switch (the effector) turns the machine on, opening up the distant active site and letting the appropriate substrate in. The enzyme then clamps down on the substrate and “reduces” it by replacing the oxygen radical with a hydrogen. When released, the DNA building block is ready for use, the effector switches the machine off, and the enzyme is ready for the next operation.
Somehow, when there are too many building blocks floating around in the cell, an effector binds to a different active site, disabling the machine. It’s uncanny how each part seems to know what’s needed and how to provide it. This involves feedback from the nucleus, where genes respond to the supply by either locking the RNR enzymes or making more of them.
Catherine Drennan on the MIT team calls this enzyme “evolutionarily ancient” and speculates about its origin.
Deoxyribonucleotides are generated from ribonucleotides, which are the building blocks for RNAs — molecules that perform many important roles in gene expression. RNR, which catalyzes the conversion of ribonucleotides to deoxyribonucleotides, is an evolutionarily ancient enzyme that may have been responsible for the conversion of the earliest life forms, which were based on RNA, into DNA-based organisms, Drennan says.
She buys into the “RNA World” scenario for the origin of life, a view that is loaded with problems. RNR is an enzyme made of protein. Did a world of floating RNA fragments somehow build this complex, multi-part protein machine before DNA-based organisms existed? That makes no sense. A ribozyme with enough “code” for RNR would itself be impossibly complex to imagine forming by chance. It would have no way to translate that code into a polypeptide without a ribosome, also made of RNA and protein. Finally, even if by multiple miracles a primitive RNR appeared with its effectors and started cranking out product, the “RNA world” would have no idea what to do with a bunch of DNA building blocks floating around. Notably, Drennan’s paper in eLife says nothing about any of this. Instead, it praises the “elegant set of protein rearrangements” performed by RNR.
RNR is one of a multitude of complex, highly-specific, multi-component machines with moving parts. Found in the simplest bacteria and archaea all the way up to human beings, it deserves better than to be treated like hopeful junk that arose by chance and found a job by accident. It deserves to be honored as an “exquisitely designed” molecular machine that performs an “intricate interplay” of functions vital to life, just like an intelligent designer would envision, plan, and create.
Image credit: Christina Zimanyi, Percival Yang-Ting Chen, and Catherine Drennan via MIT News.
