 Intelligent Design
Intelligent Design
Respect for Kinesin Grows
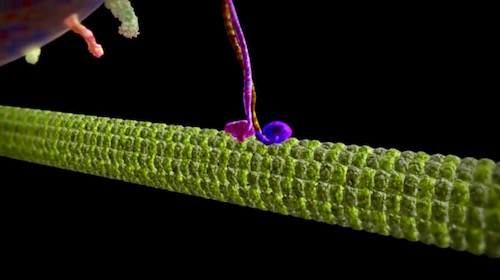
The Discovery Institute video “Workhorse of the Cell: Kinesin” premiered last year on YouTube. Since then, additional facts about these molecular motors have come to light. Some highlights:
Relay Handoff
Kinesin-II, one of the extended family of kinesin motors, is involved in a “relay race” on a micro-miniature scale. It demonstrates how different families of molecular motors cooperate. This one works in synergy with other cargo-carrying motors in the important complex function known as “intra-flagellar transport” (IFT) that builds and maintains cilia and flagella. An important example occurs in the neurons of the eye.
O’Hagan and Barr, writing for Nature Cell Reports, describe “A motor relay on ciliary tracks” between kinesin-II and another motor. It turns out that kinesin-II is the marathoner running up the neuron into the crowded transition zone (TZ), where it “hands off” its cargo a sprinter named OSM-3 that runs it up to the cilium tip.
Peterman, Scholey and colleagues have now discovered a previously unappreciated complexity in the cooperation of anterograde motors using advanced techniques ….
They find that kinesin-II acts as an import motor that transports cargos from the distal dendrite into the ciliary TZ (Fig. 1). Because the TZ is cluttered with obstacles such as Y-link attachments, kinesin-II (with its tendency to frequently detach from microtubules) may easily sidestep obstructions, switching to an adjacent microtubule protofilament to continue on its path to deliver cargo to the PS [proximal segment]. A similar model of obstacle avoidance was proposed to function in axons, in which kinesin-2 binds the same cargo as kinesin-1 to allow the complex to sidestep around Tau protein obstacles.
The authors then observed gradual acceleration of IFT in the PS, accompanied by a gradual decrease in abundance of kinesin-II and concurrent increase in OSM-3 (KIF17) (Fig. 1). The acceleration resulting from changing the ratio of kinesin-II to OSM-3 did not match previous predictions from in vitro data. Instead, a biased contribution model, in which OSM-3 outcompetes kinesin-II ten-fold, fitted the acceleration observed in vivo. These results suggested a ‘handover zone’ in the PS, in which kinesin-II gradually hands over its transport duties to the faster and more processive OSM-3, which then acts as an efficient long-range motor to bring IFT complexes to the ciliary tip. [Emphasis added.]
Noting that different kinds of cilia in various species have different families of motors acting together, the authors describe “an emerging theme” in IFT research. They sugest that “kinesin-II plays an essential role in ciliogenesis, whereas accessory motors are associated with specializations in ciliary form and function.” Many debilitating diseases are known to occur from mutations in IFT.
It’s amusing how they used the word “evolution” only one time. “Why did evolution add another anterograde motor when heterotrimeric kinesin-II seems capable of doing the job alone in algae?” As it turns out, this relay race is an efficient way to get the job done. Kinesin-II and OSM-3 each have their strengths, and each one is matched for its environment. Then, at the ciliary tip, another molecular motor — dynein — takes cargo back down.
The paper by Scholey and Peterman and colleagues in Nature Cell Biology doesn’t have anything to say about evolution. Instead, the scientists explain why “functional differentiation” provides the best way to get cargo delivered on time:
Dissociated kinesin-II motors undergo rapid turnaround and recycling to the ciliary base, whereas OSM-3 is recycled mainly to the handover zone. This reveals a functional differentiation in which the slower, less processive kinesin-II imports IFT trains into the cilium and OSM-3 drives their long-range transport, thereby optimizing cargo delivery.
Kinesin-II is no slouch as a runner. In our video, it appears to walk slowly and deliberately, but remember: the narrator says it can take a hundred steps a second. The researchers seem to like the word “optimize,” using it four times in all, such as:
We propose that the functional differentiation of kinesin-II and OSM-3 into an import motor and transport motor, respectively, optimizes sequential steps of anterograde IFT to build the cilium. The system thus exploits each kinesin-2’s specific motor properties and requires that the number of motors on the cargo, that is, the IFT train, is well orchestrated, resulting in a gradual exchange of the different motors to ensure reliable handover of cargo (Fig. 7). In a broader context, the deployment of similar, same-polarity yet functionally differentiated motors may represent a common strategy for optimizing intracellular transport.
“Optimize” is a design term. Evolutionists, by contrast, are fond of “tinkering,” wherein natural selection supposedly cobbles together parts just to get by. Which word is a better match to the observations?
Myosin Mimicry
Another walking motor is myosin, which walks along actin filaments instead of microtubules. Bioengineers at Emory University weren’t quite able to imitate its motion, but they did improve on previous attempts by inventing a DNA-based motor that rolls like a wheel. At first, the news from Emory makes it sound like they bested nature:
“Unlike other synthetic DNA-based motors, which use legs to ‘walk’ like tiny robots, ours is the first rolling DNA motor, making it far faster and more robust,” says Khalid Salaita, the Emory University chemist who led the research. “It’s like the biological equivalent of the invention of the wheel for the field of DNA machines.”
But then they have nothing but praise for natural myosin:
The field of synthetic DNA-based motors, also known as nano-walkers, is about 15 years old. Researchers are striving to duplicate the action of nature’s nano-walkers. Myosin, for example, are tiny biological mechanisms that “walk” on filaments to carry nutrients throughout the human body.
“It’s the ultimate in science fiction,” Salaita says of the quest to create tiny robots, or nano-bots, that could be programmed to do your bidding. “People have dreamed of sending in nano-bots to deliver drugs or to repair problems in the human body.”
So far, however, mankind’s efforts have fallen far short of nature’s myosin, which speeds effortlessly about its biological errands. “The ability of myosin to convert chemical energy into mechanical energy is astounding,” Salaita says. “They are the most efficient motors we know of today.”
The engineer makes this blunder in his reasoning, though:
“Our DNA-based motor can travel one centimeter in seven days, instead of 20 years, making it 1,000 times faster than the older versions,” Salaita says. “In fact, nature’s myosin motors are only 10 times faster than ours, and it took them billions of years to evolve.”
For one thing, myosins are present in the earliest cells we know of, so it didn’t take them “billions of years to evolve.” For another, if he applied intelligence to build a cheap mimic, wouldn’t that be evidence that the superior product required an intelligent cause?
Molecular Ruler
We see myosin at work in another role described in a paper in
The ability of cells to change shape underpins physiological processes from embryonic development through pathogen clearance in the immune system. The plasma membrane of cells exhibits high mechanical resistance yet permits enormous changes in cell shape. This apparent paradox is resolved by a dynamic network of actin and myosin filaments beneath the plasma membrane that forms the cortical cytoskeleton. Stress fibres, bundles of actin fibres and myosin II motors traverse the cell between focal adhesions that anchor the cell in the extracellular matrix. Actomyosin contraction against these anchors generates the force required for shape change and cell motility. Phosphorylation of regulatory myosin light chain (RMLC) stimulates myosin activity, leading to contractile force generation. The Rho-associated coiled-coil kinases (ROCK) are essential for the maintenance and integrity of stress fibres in the cell by directly and/or indirectly phosphorylating RMLC. A null mutant of the Drosophila Rok gene or deletion of ROCK1 or ROCK2 in mice results in prenatal lethality.
Their diagrams show that ROCK acts as a “molecular ruler” due to the length of its coiled coil. “This represents a new type of spatial control, and hence a new paradigm for kinase regulation.”
They try to make a case for ROCK evolving at the origin of multicellularity, but it’s little more than speculation. “Given the role of ROCK in regulating actin dynamics at focal adhesions, it is tempting to speculate that the emergence of ROCK together with the integrin-mediated signalling machinery was a crucial event in metazoan evolution.” Exactly how machinery “emerges” by blind processes is not clear. One thing is clear: it is “evolutionarily conserved throughout the animal kingdom.” In fact, it is “remarkably well conserved across more than 600 million years of evolution.” That’s hardly something to make an evolutionist cheer. We’ll take their concluding automotive analogy instead. It sounds more like design.
Our findings indicate that the activity of the Rho kinases is regulated by the spatial positioning of kinase and substrate in the cell, much like the clutch in a car engine determines whether the car is in gear or not. The engine, or kinase, is always running, but the car, or cell, doesn’t move unless the clutch, or substrate, is engaged. This represents a new paradigm in kinase-substrate regulation, whereby substrate specificity and corresponding activity are simply governed by the precise spatial positioning of enzyme and substrate.
So there you have it. Cells are inhabited by molecular motors and engines with precision parts operating under tight regulations. Now you can watch our video animation with a little more appreciation for what these amazing molecular machines do for all life, including you.
