Evolution
Evolution
 Intelligent Design
Intelligent Design
 Neuroscience & Mind
Neuroscience & Mind
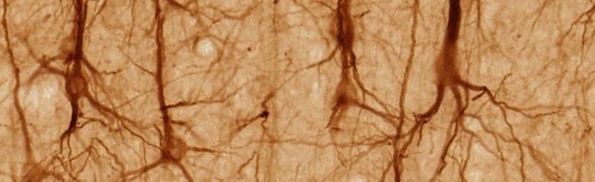
Brain Neurons Are Well Organized, Not Spaghetti Cables
They tested the randomness hypothesis. Researchers at Max Planck, Rockefeller, and Duke Universities examined the connections in brain tissue from the visual cortex, the first stop for information coming in from the retina. It was a tall order. A news item from Max Planck, “No cable spaghetti in the brain,” describes the cabling nightmare:
Nerve cells in the human brain are densely interconnected and form a seemingly impenetrable meshwork. A cubic millimeter of brain tissue contains several kilometers of wires. A fraction of this wiring might be governed by random mechanisms, because random networks could at least theoretically process information very well. Let us consider the visual system: In the retina, several million nerve cells provide information for more than 100 Million cells in the visual cortex. The visual cortex is one of the first regions of the brain to process visual information. In this brain area, various features as spatial orientation, color and size of visual stimuli are processed and represented. [Emphasis added.]
But they did not find randomness. They found a well-organized structure like a library.
The way information is sent may be comparable to a library, in which books can easier found [sic] if they are sorted not only alphabetically by title, but also by genre and by author. In a library, books are spread to different shelves, but typically not randomly. Similarly, various facets of visual perception are represented separately in the visual cortex.
Most neurons in the visual cortex behave similarly to their neighbors. Exceptions are “pinwheels” — singular points “around which the preferred orientations of the cells are arranged as the winglets of a pinwheel.” They looked to see if the number and orientation of these pinwheels was random. It was not; the observations do not fit the random hypothesis.
The scientists do not exclude the possibility that in early brain development, random connections might play a role. But through visual experience and dynamic reorganization of connections, the brain rewires itself to such a degree that only little is left from the initial wiring. “Our study shows that self-organization of neuronal circuits in the brain is the most plausible theory for the detailed structure of circuits in the visual system” explains Wolfgang Keil, who graduated from the MPI for Dynamics and Self-Organization and is currently working at Rockefeller University. This result is consistent with the observation that mammals, including us human beings, learn to see after birth. Random networks, which might exist early on, probably do not suffice for full vision.
How would this evolve by a Darwinian process? The visual cortex does not “see” the outside world. If you were a neuron, operating in the dark inside brain tissue, you would only sense chemical signals coming and going. How would neurons ever “know” how to “self-organize” in such a way that their representations of incoming signals would form a 576-megapixel motion picture that corresponds to the external world?
No mutation or series of mutations would lead to a 100-million-volume sorted library. Try getting that out of a pile of random computer cable subjected to random kicks! We know of only one cause that can organize “spaghetti cable” into a functioning network. That cause is intelligence.
Individual Neuron Guidance Cues
Meanwhile at Brown University, researchers gained insight on what guides individual neurons to connect. It’s a protein traffic cop: “The finding could help unravel the complexity of how neural connections form and help understand diseases that result from errant brain wiring.”
It’s a wonder of nature — and a darned good thing — that amid many billions of similar cells in the brain and spinal cord, neurons can extend their tendrillous axons to exactly the right place to form connections, otherwise we wouldn’t move, sense, or think properly, if at all. In a new study in the journal Science, researchers report a discovery that helps to explain how axons manage to find their way across the midline of the spinal cord.
The protein is named NELL2. It acts like a “Do Not Enter” sign, they say. “NELL2 is the first known protein to bind to a critical receptor on neurons known as Robo3,” the announcement from Brown explains. “When that binding happens, axon growth diverts away, the team’s experiments show.” But it doesn’t act alone. It’s part of a suite of proteins working together on guidance control.
NELL2 therefore joins a triad of signals that Robo3 handles in regulating axon growth. The presence of Netrin attracts axons expressing Robo3 and a particular subtype of Robo3 allows axons to ignore the repulsive signals provided by yet another protein called Slit. Expressing the Robo3 receptor therefore equips a neuron to grow its axons according to the following rules: they go where they need to (Netrin), steer clear of prohibited areas (NELL2), and ignore prohibitions meant for other neurons, but not for them (Slit).
What happens to broken cells that don’t follow the rules? Alexander Jaworski provides one example:
A rare human genetic disorder illustrates the importance of those connections. People with a mutation that prevents them from expressing Robo3 cannot move their eyes from side to side, Jaworski said. That motion is accomplished when a muscle on one side of the eye contracts and a muscle on the other side relaxes. People without Robo3 don’t have axons crossing the midline of the hindbrain and so they don’t transmit opposite signals to opposite sides of the eye. Instead, the muscles on both sides contract and the eye doesn’t move laterally.
This suggests irreducible complexity of the system. It’s not going to improve if a mutation affects one of the proteins in the triad; it’s going to break.
Interesting as these signals are, they don’t explain the larger issue: How do these proteins know when and where to signal? How can a single protein know the “big picture” that the body is aiming for during development? It’s analogous to city planners designing a computer center to control the city’s traffic signals. Somebody or something with a plan and purpose has to know the overall design goal and how to build it.
There’s no mention of evolution here, nor in the full paper in Science. The paper, however, does use a notable word: purpose. “These findings identify NELL2 as an axon guidance cue and establish Robo3 as a multifunctional regulator of pathfinding that simultaneously mediates NELL2 repulsion, inhibits Slit repulsion, and facilitates Netrin attraction to achieve a common guidance purpose.” In fact, they use it again in their concluding sentence:
Our results also show that Robo3.1 serves as an integrative hub: Its three diverse actions in response to three different cues — mediating NELL2 repulsion from the motor column, potentiating midline Netrin-1 attraction, and antagonizing midline Slit repulsion — act simultaneously, are mutually reinforcing, and serve the common purpose of steering commissural axons toward and across the midline. This multiplicity of mechanisms likely helps ensure high-fidelity steering of axons to their targets.
It’s one of those occasions in biology (not rare) when the term “intelligent design,” despite other merits, falls flat as a description. This is super-intelligent ultra-design.
Image: Pyramidal neurons in cerebral cortex, by UC Regents Davis campus [CC BY 3.0], via Wikimedia Commons.