 Evolution
Evolution
If Chemistry Can Be Wrong, How Much More Evolutionary Theory?
 Along with astronomy, chemistry is one of the ancient sciences. Progressing from alchemy to rational chemistry, physical chemistry and quantum mechanics of our day, its status as “hard science” seems secure. Its theories have been refined for centuries. Moreover, its experiments (unlike macroevolution) are observable and repeatable. How, then, could researchers at Stony Brook University (academic home, by the way, of ENV contributor Dr. Michael Egnor, Vice-Chairman, Department of Neurological Surgery) say that a discovery has challenged the foundation of chemistry?
Along with astronomy, chemistry is one of the ancient sciences. Progressing from alchemy to rational chemistry, physical chemistry and quantum mechanics of our day, its status as “hard science” seems secure. Its theories have been refined for centuries. Moreover, its experiments (unlike macroevolution) are observable and repeatable. How, then, could researchers at Stony Brook University (academic home, by the way, of ENV contributor Dr. Michael Egnor, Vice-Chairman, Department of Neurological Surgery) say that a discovery has challenged the foundation of chemistry?
Experiments at the lab are causing a stir — if not a revolution — in this hallowed science:
All good research breaks new ground, but rarely does the research unearth truths that challenge the foundation of a science. That’s what Artem R. Oganov has done, and the professor of theoretical crystallography in the Department of Geosciences will have his work published in the Dec. 20 issue of the journal Science….
“I think this work is the beginning of a revolution in chemistry,” Oganov says. “We found, at low pressures achievable in the lab, perfectly stable compounds that contradict the classical rules of chemistry. If you apply the rather modest pressure of 200,000 atmospheres — for comparison purposes, the pressure at the center of the earth is 3.6 million atmospheres — everything we know from chemistry textbooks falls apart.” [Emphasis added.]
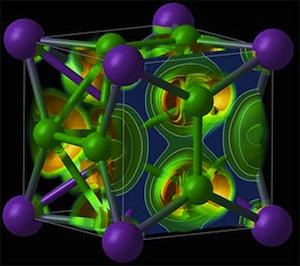
We all know NaCl, table salt. Ever heard of NaCl3? How about Na3Cl? Those are some of the novel compounds Oganov’s lab has produced. They were stunned to find them to be stable, real compounds.
“We found crazy compounds that violate textbook rules — NaCl3, NaCl7, Na3Cl2, Na2Cl, and Na3Cl,” says Weiwei Zhang, the lead author and visiting scholar at the Oganov lab and Stony Brook’s Center for Materials by Design, directed by Oganov. “These compounds are thermodynamically stable and, once made, remain indefinitely; nothing will make them fall apart. Classical chemistry forbids their very existence. Classical chemistry also says atoms try to fulfill the octet rule — elements gain or lose electrons to attain an electron configuration of the nearest noble gas, with complete outer electron shells that make them very stable. Well, here that rule is not satisfied.“
The discoveries open up new possibilities for the supposedly mature science. “When you change the theoretical underpinnings of chemistry, that’s a big deal,” one team member says. “But what it also means is that we can make new materials with exotic properties.”
How could this happen in a “hard” science? For one, Oganov, described as curious and obstinate, was driven by the word “impossible” to explore its limits.
To Oganov, impossible didn’t mean something absolute. “The rules of chemistry are not like mathematical theorems, which cannot be broken,” he says. “The rules of chemistry can be broken, because impossible only means ‘softly’ impossible! You just need to find conditions where these rules no longer hold.”
Oganov compares the team’s findings to discovering a new continent. Understanding and predicting high-pressure compounds can lead to new theories. And he envisions applications for astrophysics and planetary sciences, where high pressures abound.
If an unexpected foundation-shaking paradigm shift can occur in a “hard” science like chemistry, where findings can be checked by observation and experiment, how confident can evolutionists be that their theories about the unobservable past?
In recent years, major problems have surfaced in evolutionary theory: the overthrow of “junk DNA,” the discovery of codes within codes, the intransigence of the Cambrian enigma to name a few. Yet its advocates continue to bully anyone who doesn’t toe the line. Darwinism acts like a religion, not science. If Darwinists were proper scientists, they would embrace the new discoveries that break their rules. They would gladly follow the mounting evidence that points in a new direction for the biology of the 21st century — intelligent design.
Image credit: Electron localization function in the cubic NaCl3 structure/Stony Brook University.
