 Intelligent Design
Intelligent Design
The Spliceosome: A Dynamic Ribonucleoprotein Machine
The spliceosome has been described as one of “the most complex macromolecular machines known,” “composed of as many as 300 distinct proteins and five RNAs” (Nilsen, 2003). The animation above reveals this astonishing machine at work on the precursor mRNA, cutting out the non-coding introns and splicing together the protein-coding exons.
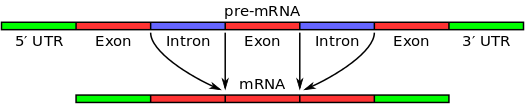
 Introns (which, unlike exons, do not code for proteins) can be of considerable length in higher eukaryotes, even spanning many thousands of bases and sometimes comprising some 90% of the precursor mRNA. In contrast, lower eukaryotes such as yeast possess fewer and shorter introns, which are typically fewer than 300 bases in length. Since introns are the non-coding segments of genes, they are removed from the mRNA before it is translated into a protein. This is not to say, of course, that introns are without important function in the cell (as I discuss here).
Introns (which, unlike exons, do not code for proteins) can be of considerable length in higher eukaryotes, even spanning many thousands of bases and sometimes comprising some 90% of the precursor mRNA. In contrast, lower eukaryotes such as yeast possess fewer and shorter introns, which are typically fewer than 300 bases in length. Since introns are the non-coding segments of genes, they are removed from the mRNA before it is translated into a protein. This is not to say, of course, that introns are without important function in the cell (as I discuss here).
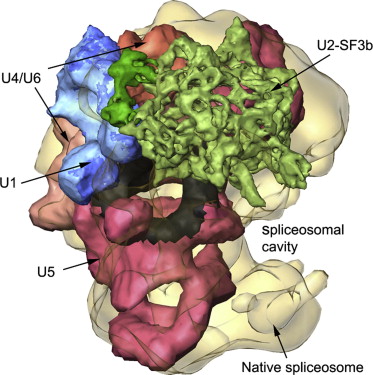
Comprising the spliceosome, shown at right (excerpted from Frankenstein et al., 2012)[1] are several small nuclear ribonucleoproteins (snRNPs) — called U1, U2, U4, U5 and U6 — each of which contains an RNA known as an snRNA (typically 100-300 nucleotides in length) — and many other proteins that each contribute to the process of splicing by recognizing sequences in the mRNA or promoting rearrangements in spliceosome conformation. The spliceosome catalyzes a reaction that results in intron removal and the “gluing” together of the protein-coding exons.
RNA Splicing
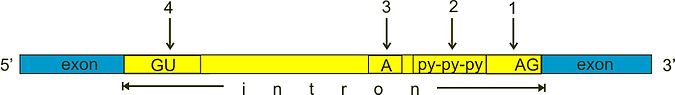
The first stage in RNA splicing is recognition by the spliceosome of splice sites between introns and exons. Key to this process are short sequence motifs. These include the 5′ and 3′ splice sites (typically a GU and AG sequence respectively); the branch point sequence (which contains a conserved adenosine important to intron removal); and the polypyrimidine tract (which is thought to recruit factors to the branch point sequence and 3′ splice site). These sequence motifs are represented in the illustration below:
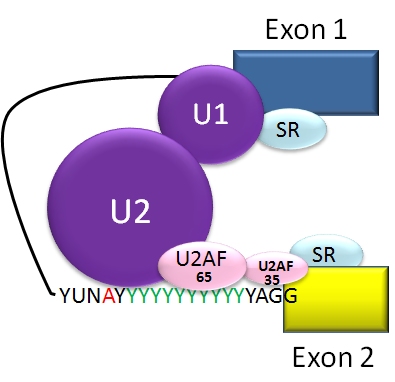
The U1 snRNP recognizes and binds to the 5′ splice site. The branch point sequence is identified and bound by the branch-point-binding protein (BBP). The 3′ splice site and polypyrimidine tract are recognized and bound by two specific components of a protein complex called U2 auxiliary factor (U2AF): U2AF35 and U2AF65 respectively.
Once these initial components have bound to their respective targets, the rest of the spliceosome assembles around them. Some of the previously bound components are displaced at this stage: For instance, the BBP is displaced by the U2 snRNP, and the U2AF complex is displaced by a complex of U4-U5-U6 snRNPs. The U1 and U4 snRNPs are also released. The first transesterification reaction then takes place, and a cut is made at the 5′ splice site and the 5′ end of the intron is subsequently connected to the conserved adenine found in the branch point sequence, forming the so-called “lariat” structure. This is followed by the second transesterification reaction which results in the splicing together of the two flanking exons. See this page for a helpful animation of the splicing process.
Other Important Protein Factors
Many other proteins play crucial roles in the RNA splicing process. One essential component is PRP8, a large protein that is located near the catalytic core of the spliceosome and that is involved in a number of critical molecular rearrangements that take place at the active site (for a review, see Grainger and Beggs, 2005). What is interesting is that this protein, though absolutely crucial to the RNA splicing machinery, bears no obvious homology to other known proteins.
The SR proteins, characterized by their serine/arginine dipeptide repeats and which are also essential, bind to the pre-mRNA and recruit other spliceosome components to the splice sites (Lin and Fu, 2007). SR proteins can be modified depending on the level of phosphorylation at their serine residues, and modulation of this phosphorylation helps to regulate their activity, and thus coordinate the splicing process (Saitoh et al., 2012; Plocinik et al., 2011; Zhong et al., 2009; Misteli et al., 1998). The illustration above (from here) shows the binding of SR proteins to splicing enhancer sites, which promotes the binding of U1 snRNP to the 5′ splice site, and U2AF protein to the polypyrimidine tract and 3′ splice site.
There are also ATPases that promote the structural rearrangements of snRNAs and release by the spliceosome of mRNA and the intron lariat. It is even thought that ATP-dependent RNA helicases play a significant role in “proofreading” of the chosen splice site, thus preventing the potentially catastrophic consequences of incorrect splicing (Yang et al., 2013; Semlow and Staley, 2012; Egecioglu and Chanfreau, 2011).
The Exon Junction Complex
The exon junction complex (EJC) is a protein complex comprised of several protein components (RNPS1, Y14, SRm160, Aly/REF and Magoh) left behind near splice junctions by the splicing process (Hir and Andersen, 2008). Their function is to mark the transcript as processed, and thus ready for export from the nucleus to the cytoplasm, and translation at the ribosome. The EJC is typically found 20 to 24 nucleotides upstream of the splice junction.
The EJC also plays an important role in nonsense mediated decay, a surveillance system used in eukaryotes to destroy transcripts containing premature stop codons (Trinkle-Mulcahy et al., 2009; Chang et al., 2007; Gehring et al., 2005). Upon encountering an EJC during translation, the ribosome displaces the complex from the mRNA. The ribosome then continues until it reaches a stop codon. If, however, the mRNA contains a stop codon before the EJC, the nonsense mediated decay pathway is triggered. The EJC and its position thus contribute to transcript quality control.
The Evolution of the Spliceosome
A popular hypothesis regarding the origins of the spliceosome is that its predecessor was self-splicing RNA introns (e.g. Valadkhan, 2007). Such a hypothesis makes sense of several observations. For example, a simpler way to achieve splicing presumably would be to bring the splice sites together in one step to directly cleave and rejoin them. The proposed scenario, however, would explain the use of a lariat intermediate, since a lariat is generated by group II RNA intron sequences (Lambowitz1 and Zimmerly, 2011; Vogel and Borner, 2002).
The hypothesis also helps to clarify why RNA molecules play such an important part in the splicing process. Examples of self-splicing RNA introns still exist today (e.g., in the nuclear rRNA genes of the ciliate Tetrahymena) (Hagen and Cech, 1999; Price et al., 1995; Price and Cech, 1988; Kruger et al., 1982).
These observations may be taken as evidence as to the spliceosome’s evolutionary predecessor, but they are hardly helpful in elucidating a plausible scenario for transitioning from one to the other. The spliceosome machinery is far more complex and sophisticated than autocatalytic ribozymes, involving not just five RNAs but hundreds of proteins.
Conclusion
The spliceosome is truly one of the most remarkable molecular machines in the cell. My purpose here was only to offer readers a small glimpse of this elegant work of nanotechnology, leaving out, of course, much important detail. As I venture deeper and deeper into the hidden world of the cell, the more I am filled with a tremendous sense of awe at the sheer genius and beauty of the design. If such engineering sophistication were encountered in any other realm of inquiry, it would immediately be attributed to intelligence. If biological systems give every appearance of having been designed, are we not justified — in the absence of a viable alternative explanation — in inferring that they most likely are the product of design
Notes:
[1] Reprinted from Structure Volume 20, Issue 6. Ziv Frankenstein, Joseph Sperling, Ruth Sperling, Miriam Eisenstein. A Unique Spatial Arrangement of the snRNPs within the Native Spliceosome Emerges from In Silico Studies. Pages 1097-1106. Copyright (2012), with permission from Elsevier.
