 Evolution
Evolution
 Intelligent Design
Intelligent Design
Our Ears Are Amazing! They Must Have Evolved
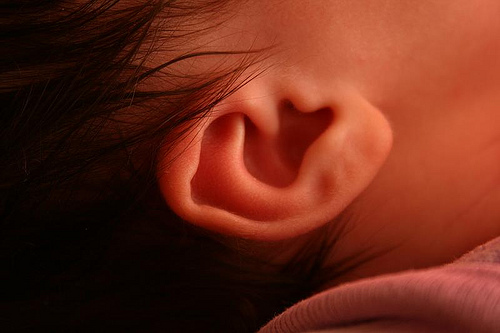
The ease with which Darwinists attribute near-perfect structures to blind evolution is astonishing. Case in point: the cochlea in the human inner ear, with its “amazing” sensitivity.
An open-access paper in PNAS by Dáibhid Ó Maoiléidigh and A. J. Hudspeth opens with a celebration of praise for the human ear:
Our ears are amazing signal detectors that reconcile great sensitivity with an enormous dynamic range. The faintest sounds that we can hear vibrate our eardrums by less than 1 pm [picometer, a trillionth of a meter] and are a trillion times less intense than the loudest sounds that we can tolerate. We can distinguish pure tones that differ in frequency by less than 0.2%, yet the frequency range of our ears exceeds a thousandfold. These features are all the more remarkable given that the mechanoreceptive organ of Corti operates in liquid and is therefore highly damped.
An active process enhances the performance of the mammalian ear by augmenting sound-induced vibrations in the cochlea. This process results from the action of specialized outer hair cells (OHCs) whose motility boosts the motion of the cochlea in response to sounds, thus amplifying the signal transmitted to the brain. These cells counteract the damping that would otherwise limit the cochlea’s sensitivity and frequency discrimination. OHCs exhibit two forms of mechanical activity, hair-bundle motility and somatic motility, which may both contribute to the cochlear amplifier. (Emphasis added.)
Wow! This is all packed into a device smaller than a snail shell. And it runs on French fries! Such astonishing, remarkable, amazing specs for a signal detector show evidence of intelligent design, right?
The authors developed a model to try to understand how hair-bundle motility (bending of the outer hair cells) and somatic motility (lengthening and shortening of the OHCs) work “synergistically” to overcome damping that would otherwise quench vibrations, especially the high frequencies. Somatic motility is mediated by millions of molecules of prestin, a protein that acts like a spring to control OHC length. By altering hair cell length and stiffness, prestin can not only tune the ear for better frequency response by minimizing damping, but can help to immediately adapt the ear’s response to loud sounds.
Most of the article deals with technical aspects such as impedance, low-pass filters, and a point of dynamic instability called a “Hopf bifurcation” where the cochlear amplifier shines. These authors (and many others) show no hesitation about speaking the jargon of acoustical engineers when dealing with this biological marvel.
But when it comes to explaining how this finely tuned, near-ideal system came into being, what do they have to say?
The results suggest that somatic motility evolved to enhance a preexisting amplifier based on active hair-bundle motility, thus allowing mammals to hear high-frequency sounds….
By virtue of their unusual cytoskeletal structure, OHCs have evidently evolved to minimize damping so that length changes owing directly to membrane-potential stimulation decrease significantly only for driving frequencies much higher than the characteristic frequency of the cell….
Active hair-bundle motility is ubiquitous among tetrapod vertebrates and likely occurs in more basal clades as well. The present model suggests an evolutionary progression in which high-frequency hearing arose when a preexisting auditory amplifier implemented by active hair-bundle motility was augmented with an additional source of mechanical energy stemming from somatic motility.
Our amazement now turns to the illogic of such explanations. They just said that “active hair-bundle motility is ubiquitous among tetrapod vertebrates and likely occurs in more basal clades as well.” This implies that the systems in basal clades are already finely tuned for high sensitivity. Where is there room for an “evolutionary progression”? How did neo-Darwinism achieve that fine-tuning in the first place? Where is the evidence of unguided evolutionary progression?
Further illogic is seen in their use of “evolve” as a teleological force. They said somatic motility “evolved to enhance.” They said OHCs “evolved to minimize damping.” They said the auditory amplifier “was augmented with an additional source of mechanical energy,” as if some dumb animal chose to do that. Neo-Darwinism does not work that way. There is no plan, purpose, or goal: just one darn mutation (mistake) after another. Those mistakes have to make it into the gametes, so that the offspring hear better. Moreover, all the offspring without the hopeful mistake have to die. Is it likely, or even credible, that a slight improvement in hearing higher frequencies could make all that much difference?
The way “evolution” is used in this and many other papers about finely tuned biological machines is a science stopper. It’s like tossing the responsibility for explanation off to a myth-teller. Thus: if it’s ideal, it evolved. If it is exquisitely sensitive, it evolved. It evolved because it evolved because it evolved. No responsibility required.
What these authors should have done is recognize what our uniform experience tells us about finely tuned, highly sensitive acoustical systems: they were designed by intelligent causes. That is the scientific explanation — not the lateral pass to the reigning myth of the day.
