 Education
Education
 Life Sciences
Life Sciences
Miller-Urey Experiment “Icon of Evolution” Alive and Well in Proposed Texas Instructional Materials

In his path-breaking book Icons of Evolution, biologist Jonathan Wells exposed how many textbooks keep recycling inaccurate evidences for chemical and biological evolution (the “icons”) long after their pull date. Unfortunately, as a Discovery Institute study documented last week, many of the “icons” cited by Wells are alive and well in supplementary instructional materials recently submitted by publishers to the Texas State Board of Education.
A prime example is the Miller-Urey origin of life experiment.
Four publishers–Apex Learning, Cengage Learning, School Education Group (McGraw Hill), and Technical Lab Systems–have proposed instructional materials for Texas that perpetuate classic errors about the famous experiment.
The Facts about Miller-Urey
Textbooks commonly claim that in the 1950s, Stanley Miller and Harold Urey performed experiments showing how the “building blocks” of life, such as amino acids, could have arisen on the early Earth. Textbooks typically show a diagram of the glass apparatus used by Miller and Urey to purportedly simulate lightning strikes hitting the earth’s early atmosphere. The claim is usually made that the experiments accurately simulated early earth conditions by using the gasses methane and ammonia to represent the earth’s early atmosphere.
However, it has been known for decades that the Earth’s early atmosphere probably was not composed of methane or ammonia, and thus would not have been conducive to Miller-Urey type chemistry. As origin of life theorist David Deamer explains, “This optimistic picture began to change in the late 1970s, when it became increasingly clear that the early atmosphere was probably volcanic in origin and composition, composed largely of carbon dioxide and nitrogen rather than the mixture of reducing gases assumed by the Miller-Urey model. Carbon dioxide does not support the rich array of synthetic pathways leading to possible monomers…”1 Theorist Jeffrey Bada and other experts have likewise observed that “Geoscientists today doubt that the primitive atmosphere had the highly reducing composition Miller used…”2
There are strong reasons to expect that the early earth’s atmosphere did not contain significant amounts of methane, ammonia, or high concentrations of other reducing gasses. The earth’s early atmosphere is thought to have been produced by outgassing from volcanoes on the early earth, and the composition of those volcanic gasses is related to the chemical properties of the earth’s inner mantle. Multiple studies have found that the chemical properties of the earth’s mantle would have been the same in the past as they are today.3 But today, volcanic gasses do not contain methane or ammonia, and are not reducing. A paper in Earth and Planetary Science Letters found that these chemical properties have been essentially constant over earth’s history, leading to the conclusion that “Life may have found its origins in other environments or by other mechanisms.”4
The Miller-Urey experiment is typically cited to lend plausibility to a “primordial soup” hypothesis, and the unguided chemical origins of life. But many leading theorists today have abandoned the Miller-Urey experiment and the “primordial soup” theory it is claimed to support. In February 2010, NPR reported that biochemist Nick Lane believes that the primordial soup theory is “past its expiration date.”5 So drastic is the evidence against pre-biotic synthesis of biological monomers that in 1990 the Space Studies Board of the National Research Council recommended that origin of life scientists undertake a “reexamination of biological monomer synthesis under primitive Earthlike environments, as revealed in current models of the early Earth.”6
Unfortunately, textbooks rarely inform students that the Miller-Urey experiments probably did not accurately model the early earth, or that leading theorists no longer consider the Miller-Urey experiments as a viable explanation for a “primordial soup.”
What Do Proposed Supplemental Materials in Texas Say?
As noted, at least four publishers have proposed instructional materials to the Texas State Board of Education that present inaccurate, misleading, or one-sided information about the Miller-Urey experiment. By doing so, these publishers have failed to follow state science curriculum standards, which call for materials that help students “analyze, evaluate, and critique scientific explanations by using empirical evidence, logical reasoning, and experimental and observational testing, including examining all sides of scientific evidence of those scientific explanations, so as to encourage critical thinking by the student.”
Following is a run-down of the inaccurate information presented:
Technical Lab Systems
The Technical Lab Systems curriculum wrongly states: “In this experiment steam was generated and passed through gasses similar to the atmosphere of the early Earth.” (emphasis added)
It then asks a question which forces students to assent to the inaccurate claim that the experiment accurately simulated conditions on the early earth:

Needless to say, there is no critique of the Miller-Urey experiment nor evaluation of its claims; and there is no presentation here of all sides of the evidence as required by Texas’s science curriculum standards.
Cengage Learning
The Cengage Learning curriculum overstates and misstates the results of the Miller-Urey experiment, declaring: “With energy provided by sunlight, lightning, and the heat of volcanoes, those compounds apparently came together to form amino acids. Those amino acids, in turn, reacted with each other to form proteins, the building blocks of all forms of life. In 1953, American chemist Stanley Miller (1930-) showed in a laboratory experiment how such reactions might take place.”
Stanley Miller died in 2007, so this text wrongly implies he is still alive. More importantly, the passage misrepresents Miller’s experiments by claiming that they showed how organic molecules could arise on the early Earth. There is no objective evaluation, analysis, critique, or presentation of all sides of the evidence pertaining to this claim.
Apex Learning
In a section titled “4.3.1 Life on Earth, The Formation of Biological Molecules,” this proposed instructional material states regarding the Miller-Urey experiment: “They found that when gases that existed in the Earth’s early atmosphere were subjected to continuous, high amounts of energy under certain chemical conditions, amino acids were formed.” As seen in the diagram below, the Apex curriculum wrongly implies that the early Earth’s atmosphere contained appreciable amounts of ammonia and methane:
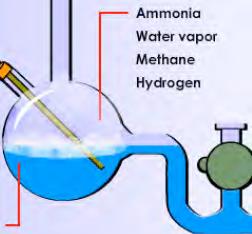
The Apex curriculum concludes that “It may have been possible for organic molecules to form spontaneously in primitive oceans.” However, there is no evaluation or critique or presentation of all sides of the data pertaining to this claim.
School Education Group (McGraw Hill)
As seen in the diagram below, this curriculum inaccurately implies that the Miller-Urey experiment “simulated early Earth environments.”

There is no evaluation or critique of these claims, nor is there any presentation of all sides of the evidence.
How Long Will Students Be Misled?
The big question now is whether the Texas State Board of Education will force publishers to correct this misinformation. More than a decade after Icons of Evolution–and more than a half-century after Miller-Urey–isn’t it time for students to get an accurate description of the facts about this 1950s experiment?
References Cited:
[1.] David W. Deamer, “The First Living Systems: a Bioenergetic Perspective,” Microbiology & Molecular Biology Reviews, Vol. 61:239 (1997).
[2.] Adam P. Johnson, H. James Cleaves, Jason P. Dworkin, Daniel P. Glavin, Antonio Lazcano, Jeffrey L. Bada, “The Miller Volcanic Spark Discharge Experiment,” Science, Vol. 322:404-405 (October 17, 2008).
[3.] Kevin Zahnle, Laura Schaefer, and Bruce Fegley, “Earth’s Earliest Atmospheres,” Cold Spring Harbor Perspectives in Biology (2010) (“Geochemical evidence in Earth’s oldest igneous rocks indicates that the redox state of the Earth’s mantle has not changed over the past 3.8 Gyr”) (internal citations omitted).
[4.] Dante Canil, “Vanadian in peridotites, mantle redox and tectonic environments: Archean to present,” Earth and Planetary Science Letters, Vol. 195:75-90 (2002) (internal citation removed) (“Abiotic synthesis of molecules and hydrocarbons that can lead to life in early Archean mantle-derived volcanic gases requires they contain significant H2 and CO, but such reduced components are not supported by results of this and many other studies, which imply a scenario of Archean mantle redox not unlike that of today. Life may have found its origins in other environments or by other mechanisms.”) (emphasis added).
[5.] Deborah Kelley, “Is It Time To Throw Out ‘Primordial Soup’ Theory?,” NPR (February 7, 2010).
[6.] National Research Council Space Studies Board, The Search for Life’s Origins (National Academy Press: Washington D.C., 1990).
